|
The relationship given by Planck's radiation law, given below, shows that with increasing temperature, the total radiated energy of a body increases and the peak of the emitted spectrum shifts to shorter wavelengths. This dependence has also been suggested previously by Hartnagel and Kirchartz. The corresponding intensity dependence of the photocurrent is described by 1 1 / ( 2 + 2 l). While Planck originally regarded the hypothesis of dividing energy into increments as a mathematical artifice, introduced merely to get the correct answer, other physicists including Albert Einstein built on his work, and Planck's insight is now recognized to be of fundamental importance to quantum theory.Įvery physical body spontaneously and continuously emits electromagnetic radiation and the spectral radiance of a body, B ν, describes the spectral emissive power per unit area, per unit solid angle and per unit frequency for particular radiation frequencies. For exponentially distributed tail states, we thus expect S to vary between S 1 / 4 and S 0 in this case, depending on E U. In 1900, German physicist Max Planck heuristically derived a formula for the observed spectrum by assuming that a hypothetical electrically charged oscillator in a cavity that contained black-body radiation could only change its energy in a minimal increment, E, that was proportional to the frequency of its associated electromagnetic wave. Īt the end of the 19th century, physicists were unable to explain why the observed spectrum of black-body radiation, which by then had been accurately measured, diverged significantly at higher frequencies from that predicted by existing theories. In physics, Planck's law (also Planck radiation law : 1305 ) describes the spectral density of electromagnetic radiation emitted by a black body in thermal equilibrium at a given temperature T, when there is no net flow of matter or energy between the body and its environment. Our eyes as well as most light energy measuring instruments cannot respond this fast and only an average energy over many cycles is observed. The classical (black) curve diverges from observed intensity at high frequencies (short wavelengths). I p 0 r r 2 E 2 c 0 n 2 E 2 where v p is the phase velocity, c is the vacuum velocity of light, and n is the refractive index. Shown here are a family of curves for different temperatures. 
The light energy at twice the distance away is spread over four times the area. Planck's law accurately describes black-body radiation. photons of light become spread over a wider area. check the math derived above) by reading the slit separation in the control box, dragging measuring devices from above the control box, and pausing the simulation. You can even explore this phenomenon quantitatively (i.e. Clearly, the larger the strength of the electric and magnetic fields, the more work they can do and the greater the energy the electromagnetic wave carries.Ī wave’s energy is proportional to its amplitude squared ( circuit containing a 1.00-pF capacitor oscillates at such a frequency that it radiates at a 300-nm wavelength.Not to be confused with Planck relation or Planck's principle. You can click on the intensity toggle box in the control box to see the graph of the intensity at the screen, as described by Equation 3.2.8. If absorbed, the field strengths are diminished and anything left travels on. Once created, the fields carry energy away from a source. With electromagnetic waves, larger E-fields and B-fields exert larger forces and can do more work.īut there is energy in an electromagnetic wave, whether it is absorbed or not. Energy carried by a wave is proportional to its amplitude squared. This simultaneous sharing of wave and particle properties for all submicroscopic entities is one of the great symmetries in nature. These particle characteristics will be used to explain more of the properties of the electromagnetic spectrum and to introduce the formal study of modern physics.Īnother startling discovery of modern physics is that particles, such as electrons and protons, exhibit wave characteristics. But we shall find in later modules that at high frequencies, electromagnetic radiation also exhibits particle characteristics. You can also measure intensity and then assume a spectrum (e.g. Luminous intensity is defined as dId / d, where d is the luminous flux (light energy flux in watts per m2) emitted within a solid angle d. If you are ok with measuring intensity, then you can do that, although an LDR is a poor choice. I have been surfing the internet and my physics books for the relationship between the resistance of an LDR and light intensity (a formula), and so far I have found nothing. The formula for this is: light intensity 1/distance. There is no way to do that with a LDR, so no this is not possible. Light intensity is inversely proportional to the square of the distance. 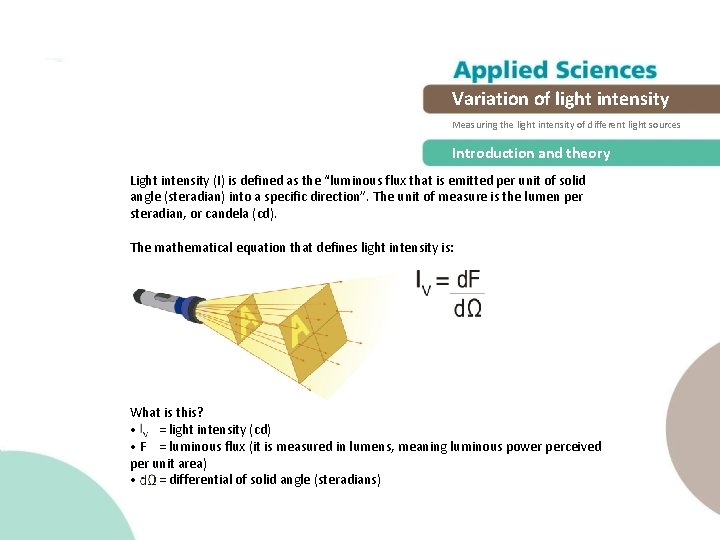
The behavior of electromagnetic radiation clearly exhibits wave characteristics. Measuring lux requires you to measure the spectrum of a light source.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
